Core Competencies & Quality
Accuracy, professionalism, and efficiency are exactly what you can expect from us. As evidence of our efforts, our facilities have obtained ISO13485 certification and our products have received CE0197 certification. It allows our medical goods to meet standards approved all over the world: South Asia, Central Asia, Middle East, Europe, South, and Central America as well as Africa. We deliver the highest quality service and products with professionalism and knowledge.
Main Product Range:
- Infusion and Injection Systems
- Respiratory Anesthesia and Emergency Care
- Disposable Catheter and Tube
- Urology and Drainage Products
- Hemodialysis and Blood Collection Products
- Gynecology and Pediatric Products
- Surgical and Diagnostic Products
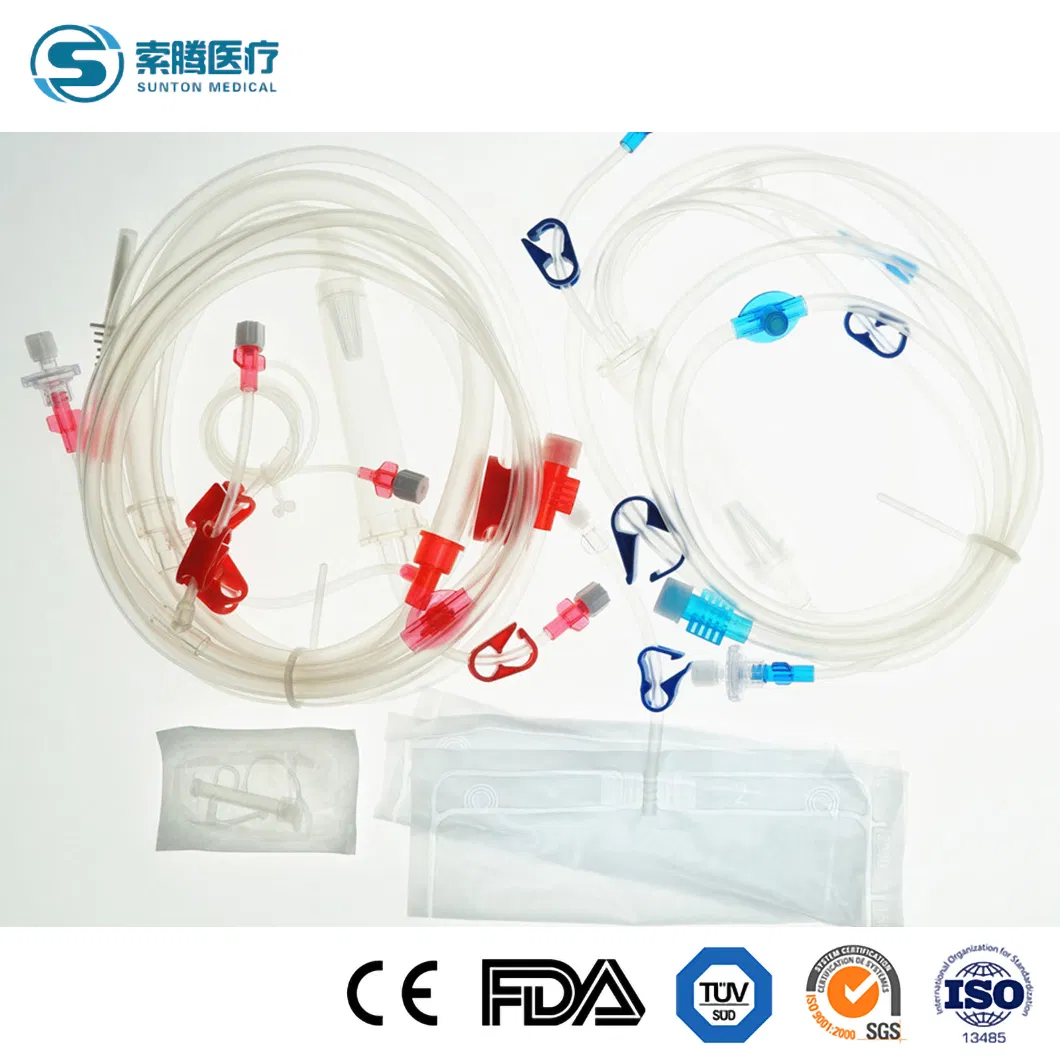
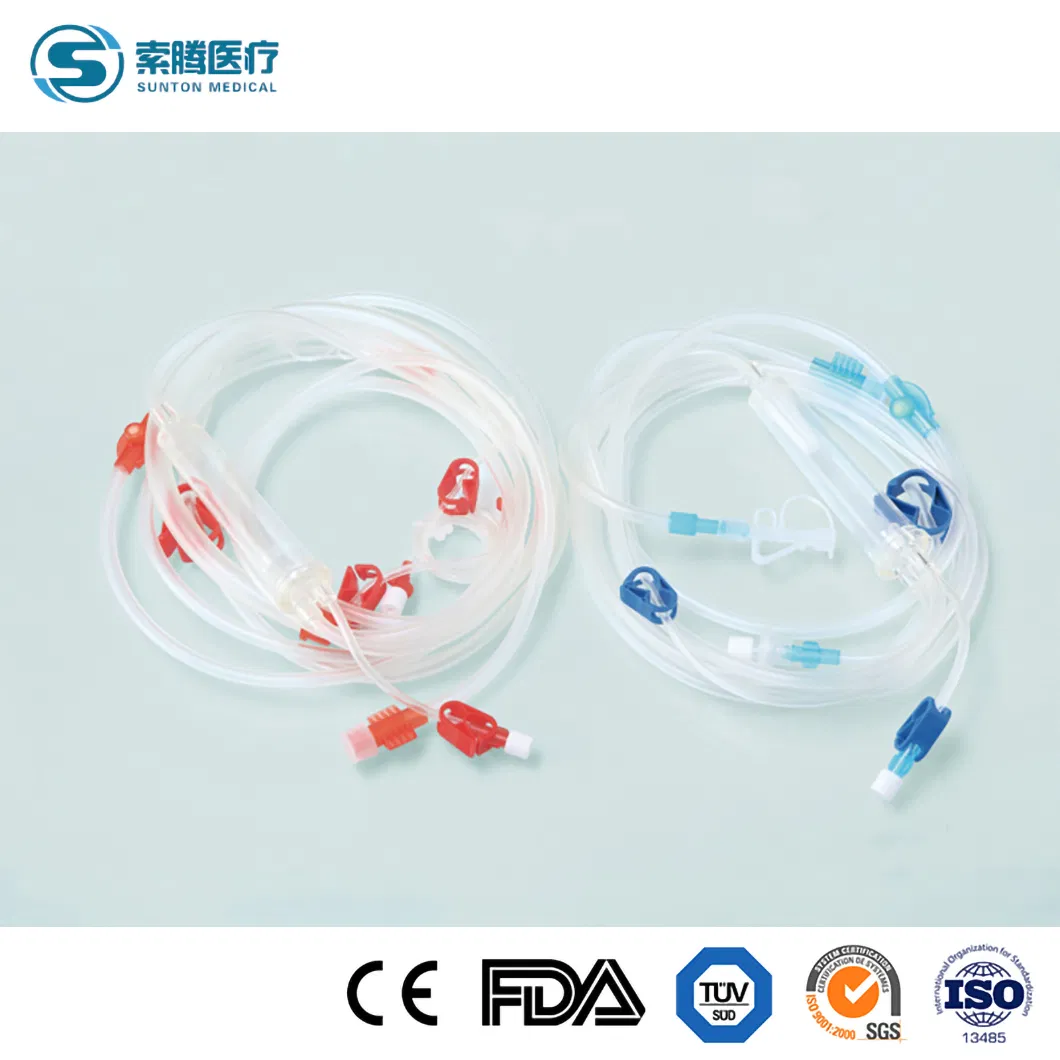
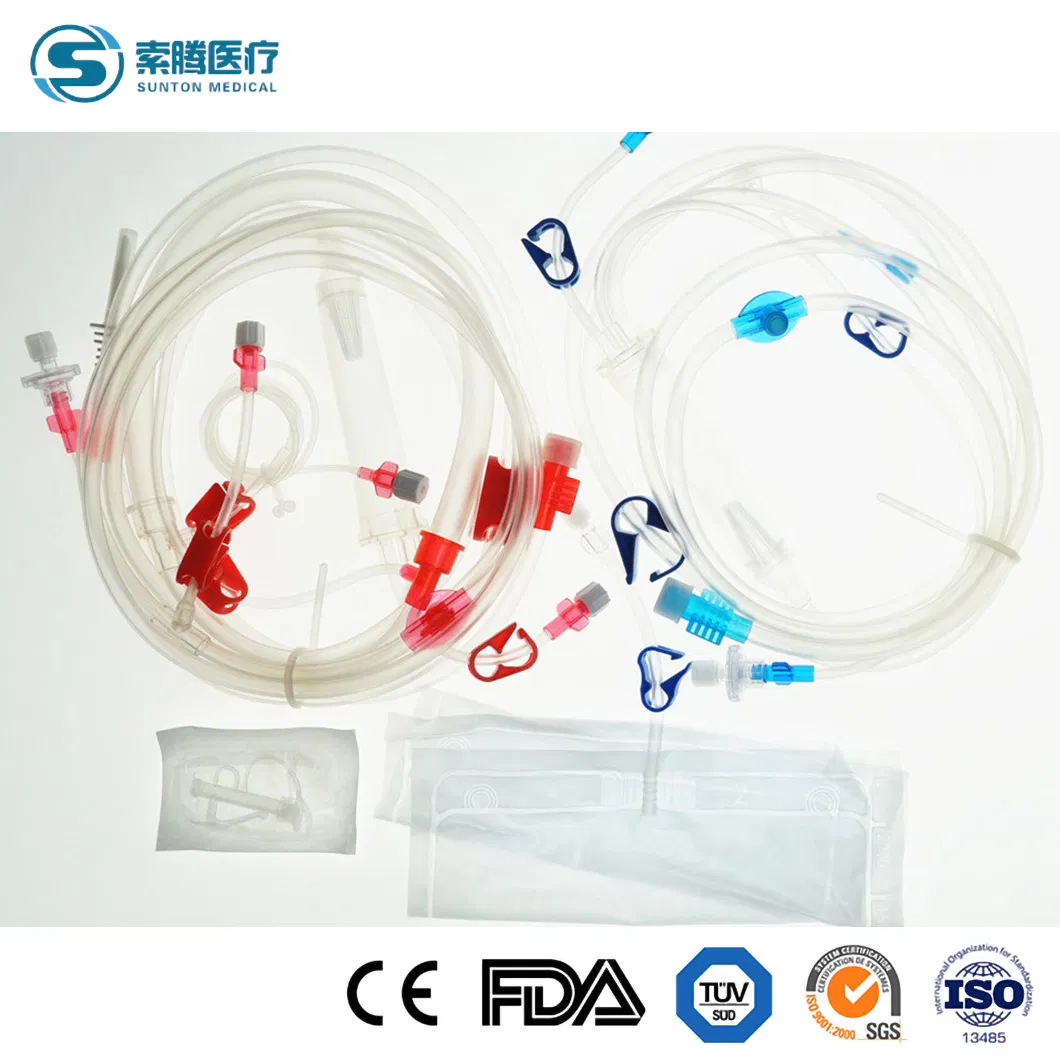
Disposable Hemodialysis Blood Lines
Disposable bloodlines connect with other medical devices to establish blood circulating paths outside the human body in the process of hemodialysis. The material of this product is a medical-grade polymer. The tubes are soft and transparent, ensuring convenient and credible connection with fistula needles and dialyzers.
⚠️ Caution:
1. Prior to use read instructions.
2. Do not store in sunlight, high humidity, or low-temperature circumstances.
3. Single-use only. Treat as infectious medical waste after use.
4. Handle under asepsis circumstances when connecting to dialyzer.
5. Sterilized with Ethylene Oxide (No pyrogen). Do not use if package is damaged.
6. If blood leaks, replace entire bloodlines immediately.
7. Pressure Monitoring Tube must connect with Transducer Protector before use.
Frequently Asked Questions
Q1: What are disposable hemodialysis blood lines used for?
They are used to connect medical devices to establish blood circulating paths outside the human body during the hemodialysis process.
Q2: What materials are used in these blood lines?
The products are manufactured using medical-grade PVC and high-quality polymers, ensuring softness, transparency, and durability.
Q3: Are these products certified for international use?
Yes, our facilities are ISO13485 certified, and our products carry CE0197 certification, meeting global medical standards.
Q4: How is sterilization handled?
All blood lines are sterilized using Ethylene Oxide (EOS) and are guaranteed to be non-pyrogenic if the package remains undamaged.
Q5: Can these blood lines be reused?
No, these are strictly single-use medical devices. They should be treated as infectious medical waste after use.
Q6: What services are provided regarding shipping and payments?
We accept delivery terms including FOB, CFR, CIF, and DDP. Payment options include T/T, L/C, MoneyGram, PayPal, and Western Union.