1 / 5










| Model NO. | Fr6-Fr16 |
| Material | PVC |
| Feature | Reusable |
| Certification | CE, FDA, ISO13485 |
| Group | Children |
| Inner Package | Blister Individually |
| OEM Service | Yes |
| Color | Transparent |
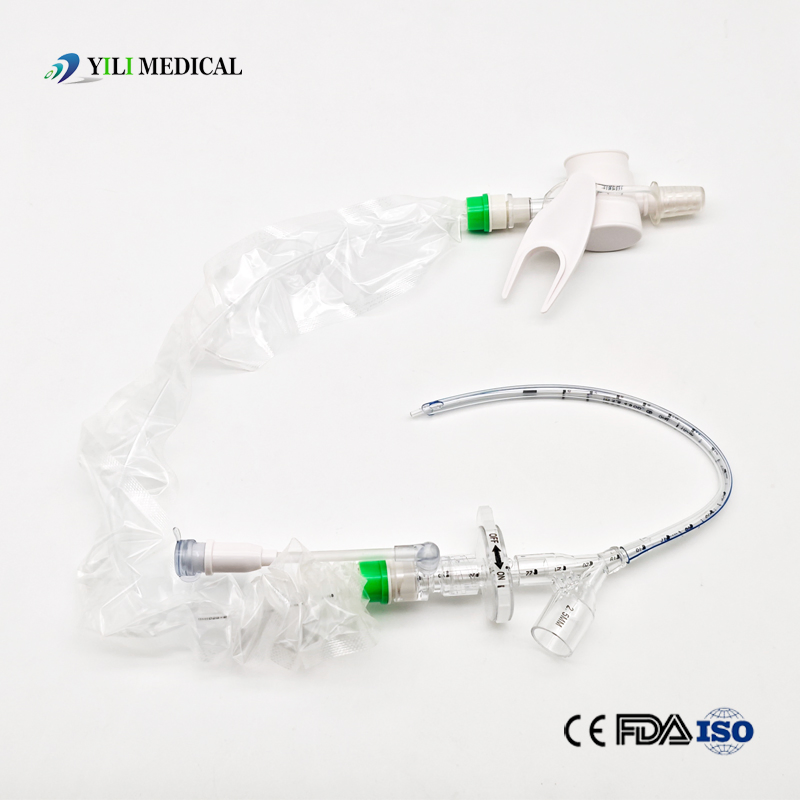
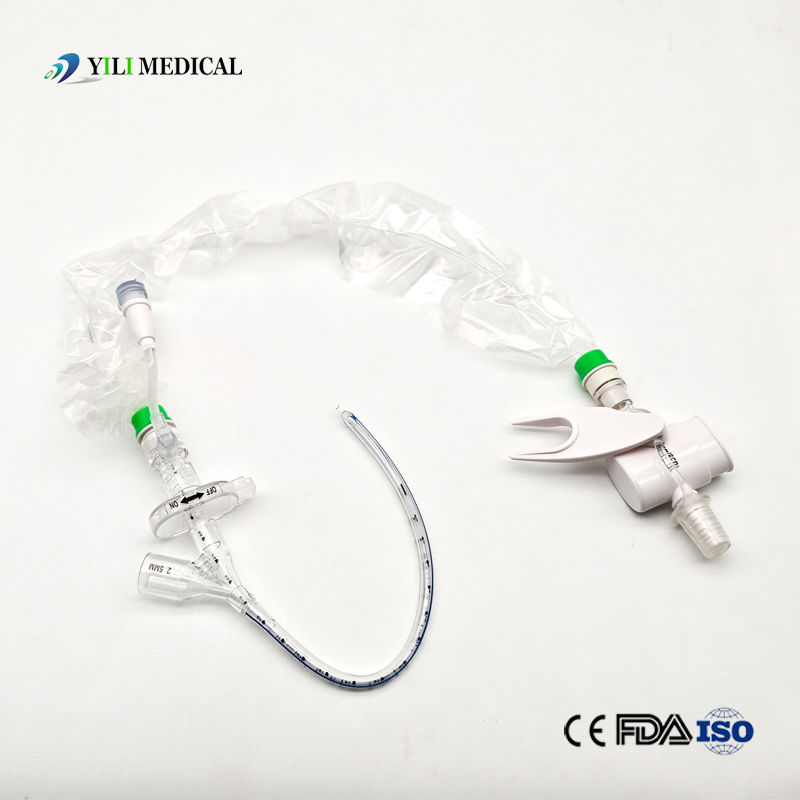
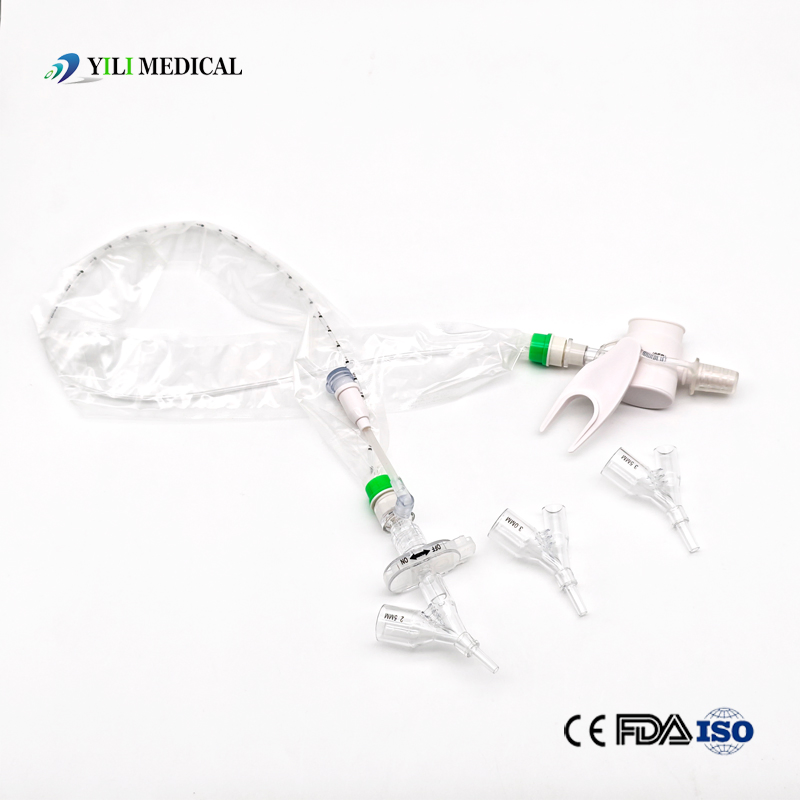
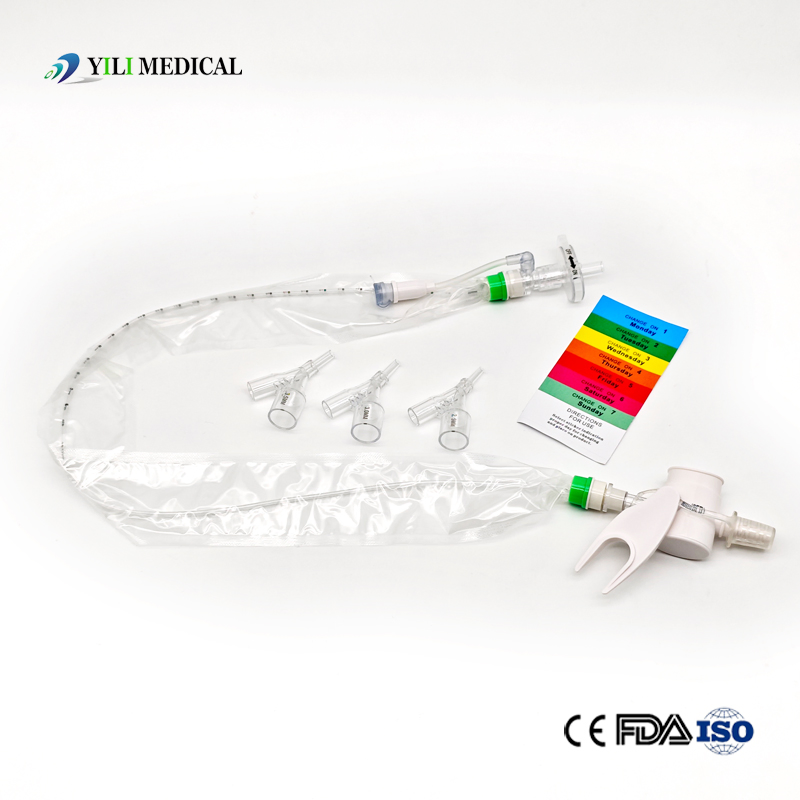
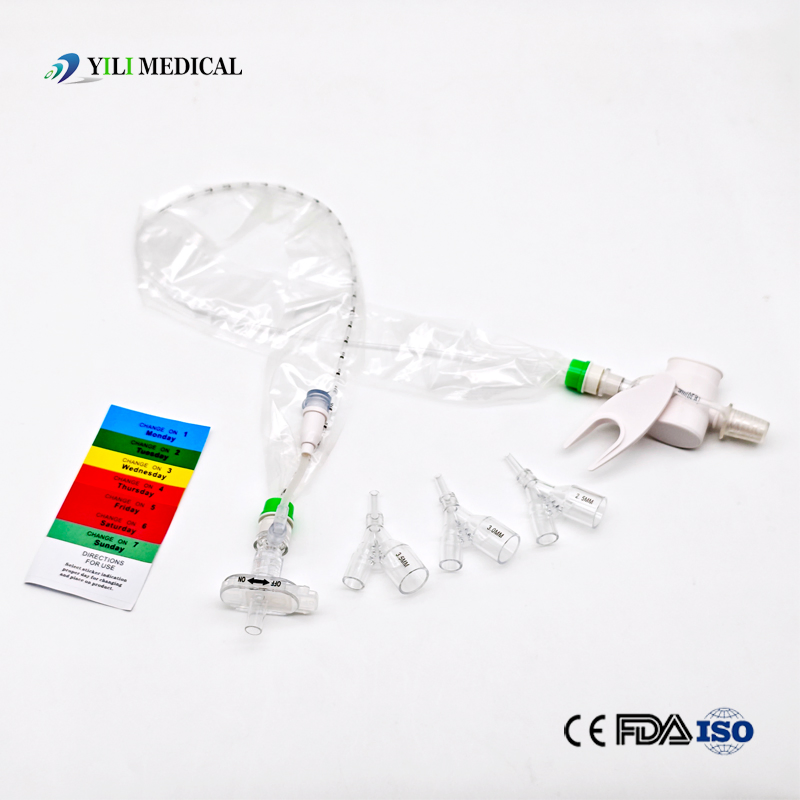
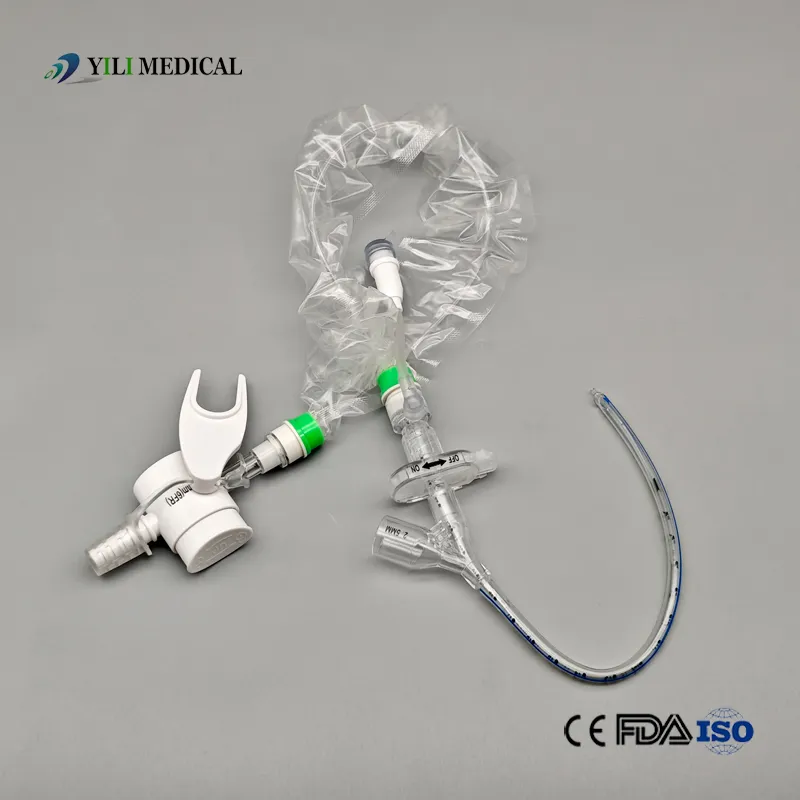
| Name | Closed Suction Catheter |
| HS Code | 9018390000 |
| Production Capacity | 5,000,000 PCS/Month |
What is a Closed Suction System?
The Closed Suction System is intended for removal of secretion in the lower respiratory track in patients receiving prolonged mechanical ventilation. The filter-protected system is connected to the artificial airway of the patient (Endotracheal or Tracheostomy tube) and to the ventilation circuit. It is used over a period of 24 hours to 72 hours, depending on the patient requirement.
| Product Attributes | |
| Application | Personal Care / ICU Support |
| Properties | Medical Materials & Accessories |
| After-sale Service | Online technical support |
Advantages of Using Closed Suction System:
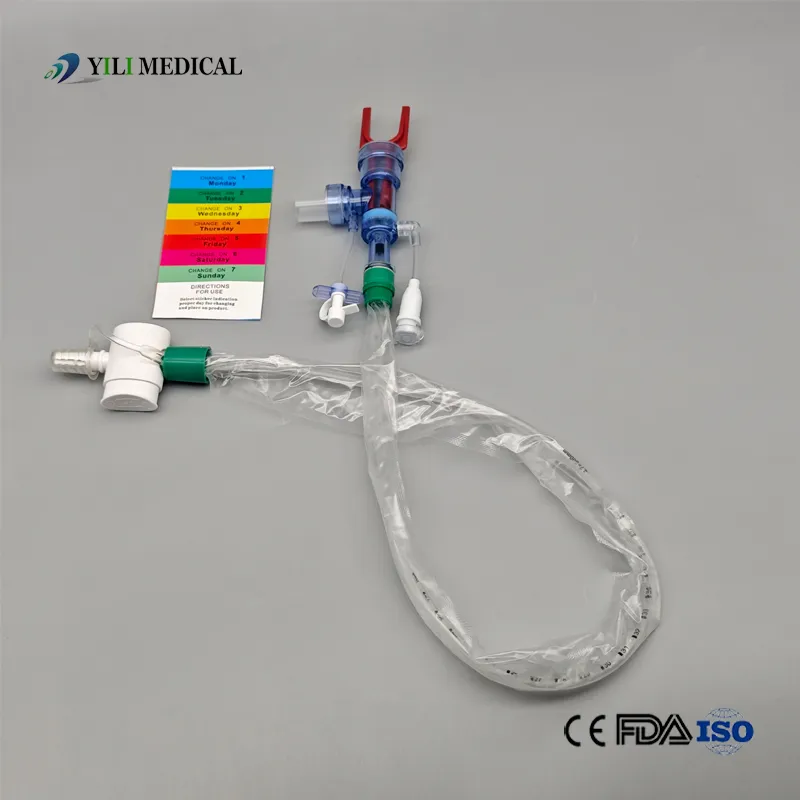
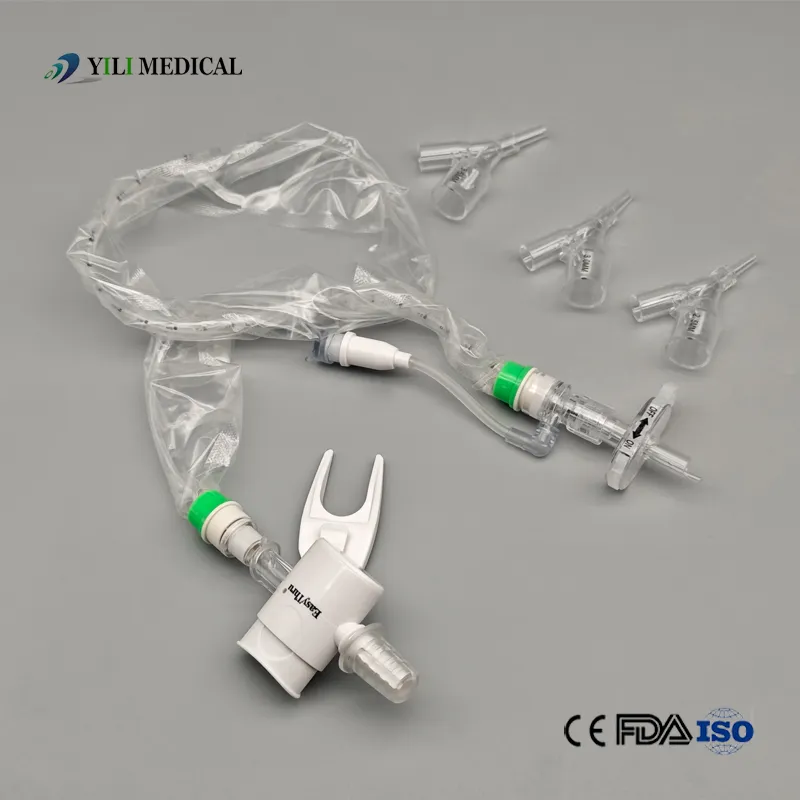
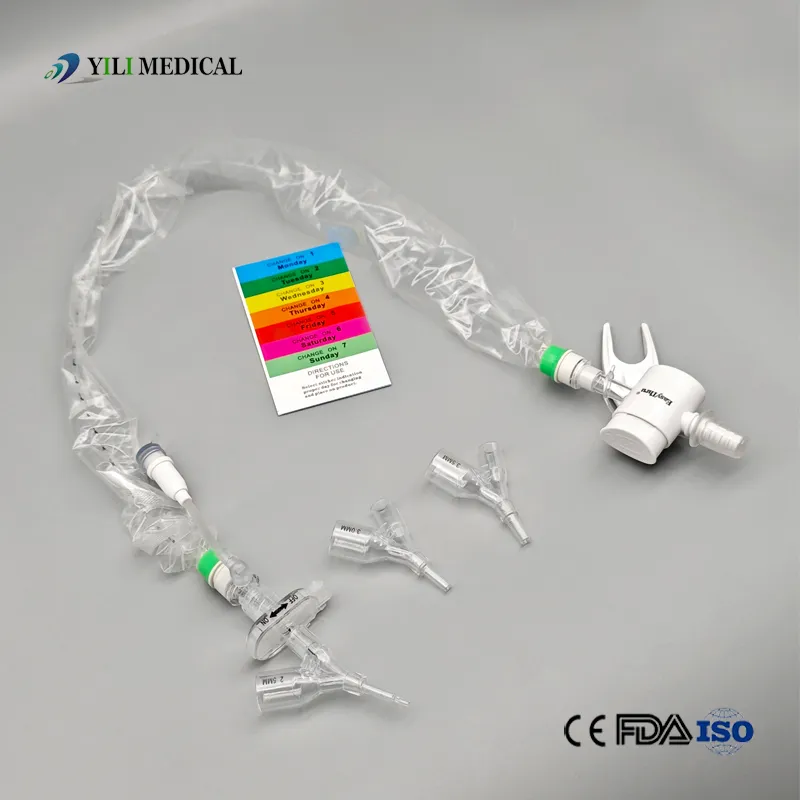


Product Qualification:





