Basic Information
Sterilization
Ethylene Oxide Sterilization
Guarantee Period
Five Years
Logo Printing
Without Logo Printing
Transport Package
Poly Bag
Product Overview
About Our Manufacturing:
We are an ISO13485 certified manufacturing factory specializing in tooling and molding for medical products.
In the past ten years, we have made and exported thousands of molds to over 30 countries around the world. These molds cover various fields, including:
- Infusion and Transfusion Systems
- Disposable Syringes
- Hemodialysis Components
- Respiratory Equipment
- Surgical Instruments and Diagnostic Equipment
In addition to mold development, we offer Plastic Injection Molding in an ISO Class 8 clean room environment. The OEM molding is typically for plastic parts of Class III medical devices, such as Catheter and Delivery System components.
Advanced Tool Design
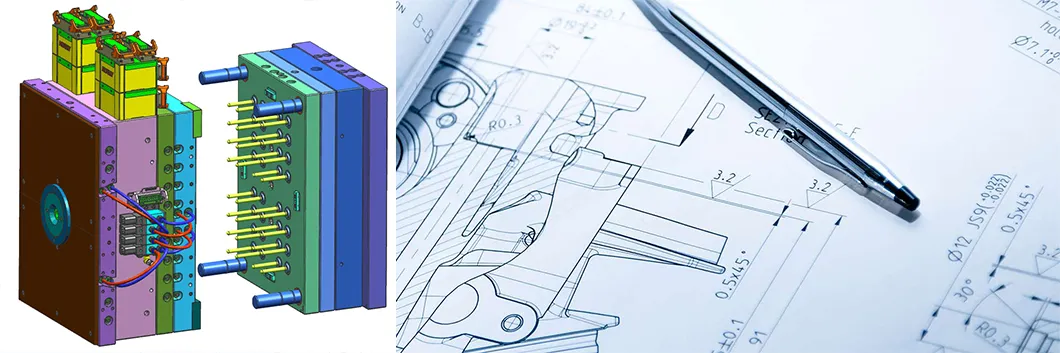
Design for manufacturability and efficient production is our primary objective. With advanced CAD/CAM/CAE systems, we provide comprehensive design and analysis.
Supported File Formats:
STEP, PRT, SLDPRT, X_T, DXF, etc.
Our sophisticated designing team with decades of experience ensures the most appropriate manufacturing options for your product, complying with all production capacity requirements.
We have rich experience in various medical markets, including INFUSION SET, IV CATHETER, DISPOSABLE SYRINGES, HEMODIALYSIS BLOODLINE, 3 WAY STOPCOCK, and DIAGNOSTIC DEVICES.
International Presence
Every year we attend international medical exhibitions to showcase our latest innovations and connect with global partners.
Quality Management System
Our facility operates under strict quality control standards to ensure the highest level of safety for medical device components.
Frequently Asked Questions
Q1: What sterilization method is used for these components?
All products undergo Ethylene Oxide (EO) Sterilization to meet medical safety standards.
Q2: What is the quality guarantee period for the products?
We provide a comprehensive five-year quality guarantee period for our medical-grade components.
Q3: In what environment are the plastic parts manufactured?
Our plastic injection molding is performed within an ISO Class 8 clean room environment, specifically designed for Class III medical devices.
Q4: Which medical fields do your molding services cover?
Our expertise spans across Infusion/Transfusion, Syringes, Hemodialysis, Respiratory, surgical instruments, and Diagnostic equipment.
Q5: What design software and file formats do you support?
We use advanced systems like Pro-E, UG, and AutoCAD. We can work with STEP, PRT, SLDPRT, X_T, and DXF file formats.
Q6: Do you participate in global medical exhibitions?
Yes, we regularly attend major international exhibitions including CMEF, MEDICA, Arab Health, and FIME.