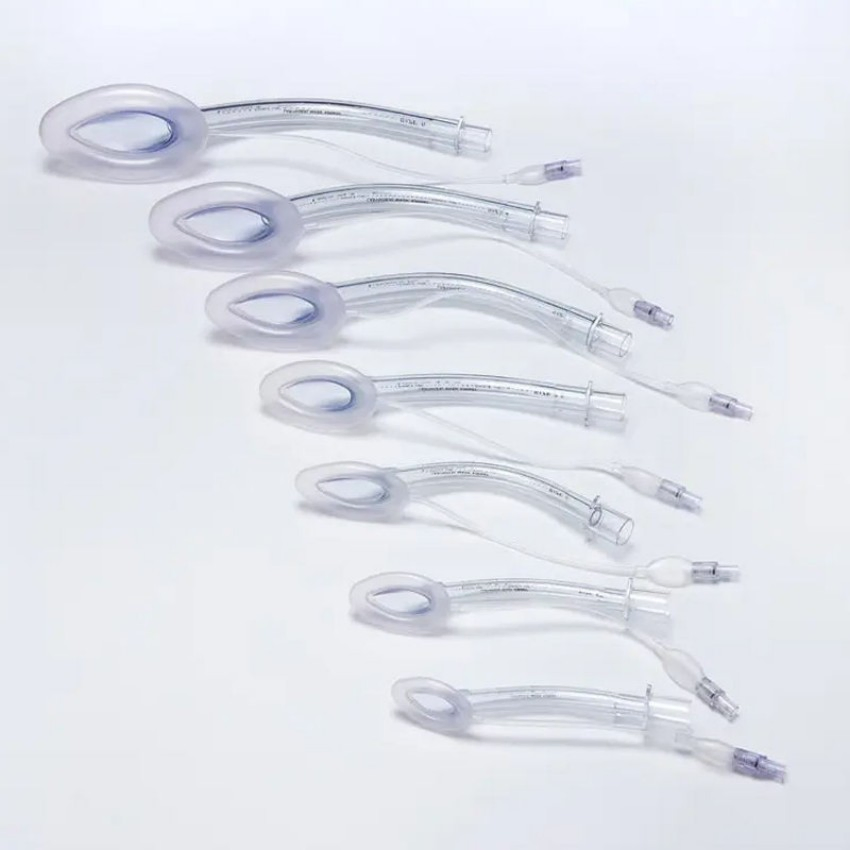


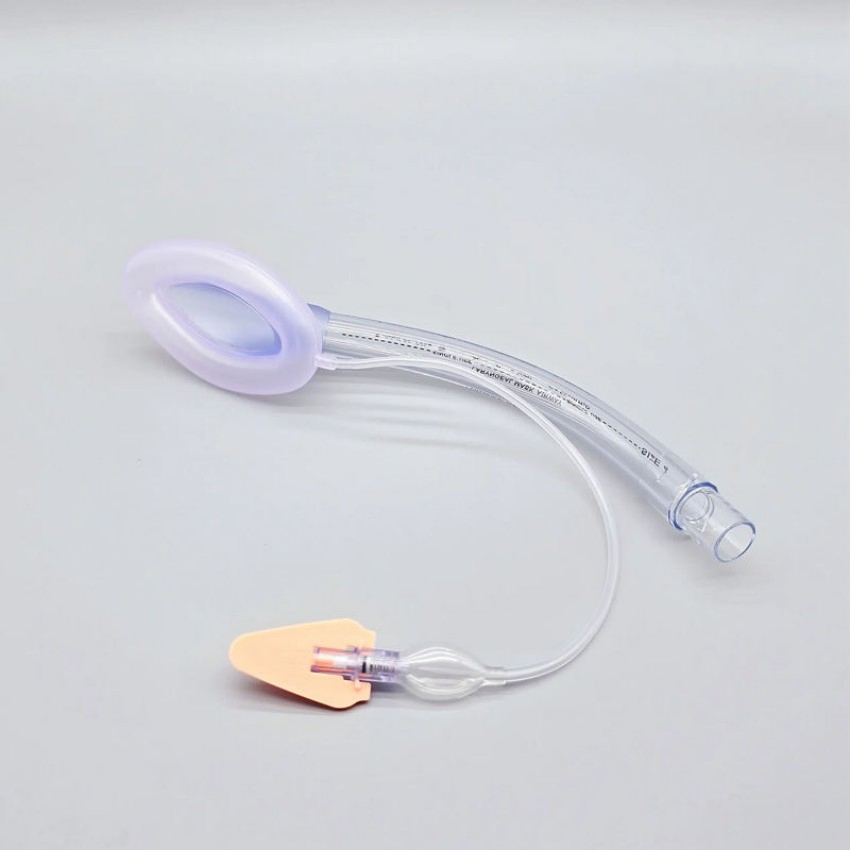










Fiberoptic Ready LMAs for Difficult Airway Management and Guided Intubation
The Advanced Supraglottic Airway Device (SAD) is engineered to provide a secure and efficient airway solution for anesthesia and emergency airway management. Manufactured from high-quality medical-grade silicone or PVC, it offers superior sealing performance, reduced airway trauma, and easy insertion - ensuring patient safety and clinician convenience.
This supraglottic airway system is ideal for short-term ventilation during surgical procedures or in emergency settings where endotracheal intubation is not possible or necessary.
| Item | Description |
|---|---|
| Product Name | Advanced Supraglottic Airway Device |
| Material | Medical-grade Silicone / PVC |
| Type | Reusable or Disposable |
| Size Options | 1#, 1.5#, 2#, 2.5#, 3#, 4#, 5# (Neonates to Adults) |
| Connector | Standard 15mm ISO Connector |
| Cuff Type | Inflatable Cuff / Non-inflatable Cuff (optional) |
| Sterilization | EO Gas Sterilized (Single Use) or Autoclavable (Reusable Type) |
| Shelf Life | 3-5 Years |
| Packaging | Individual Blister Pack |
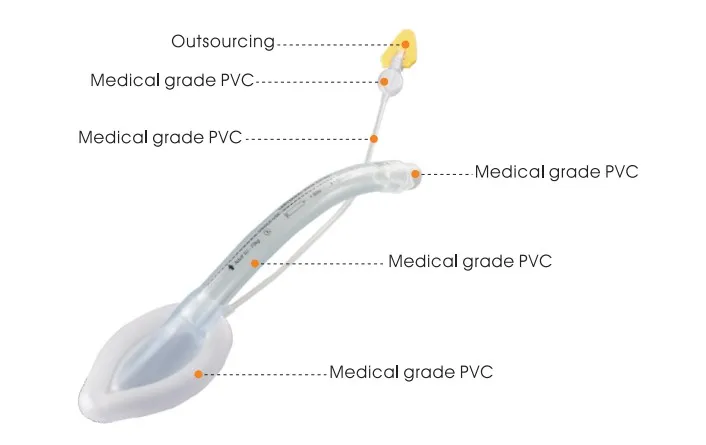
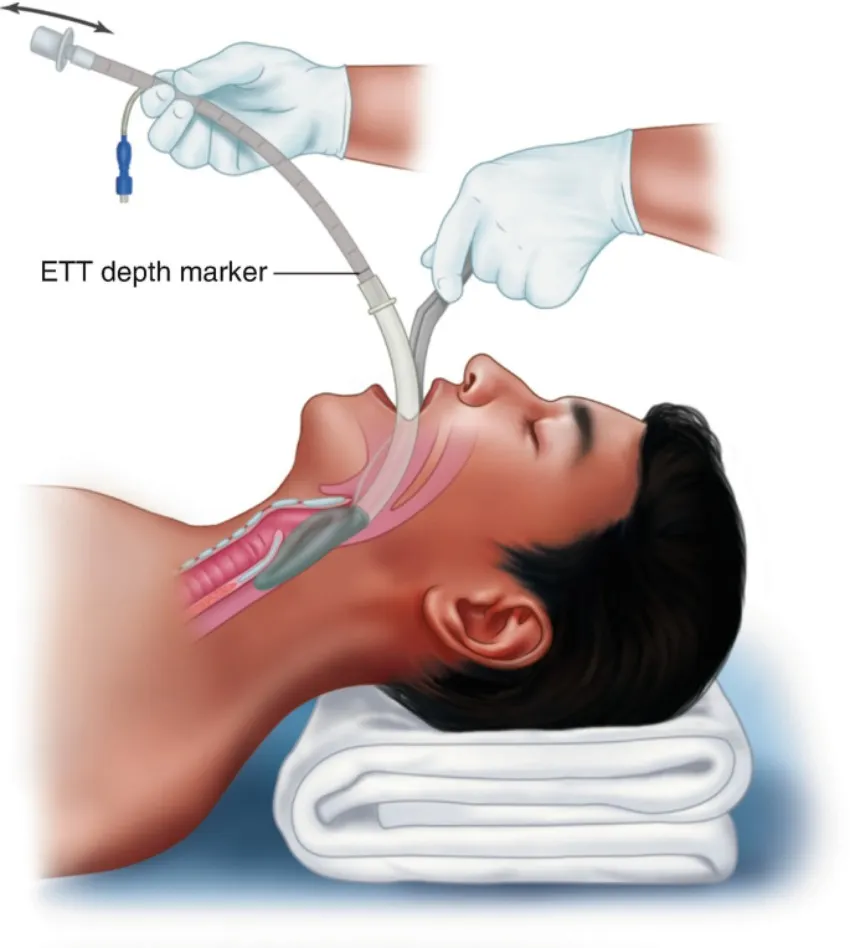

Key Advantages:
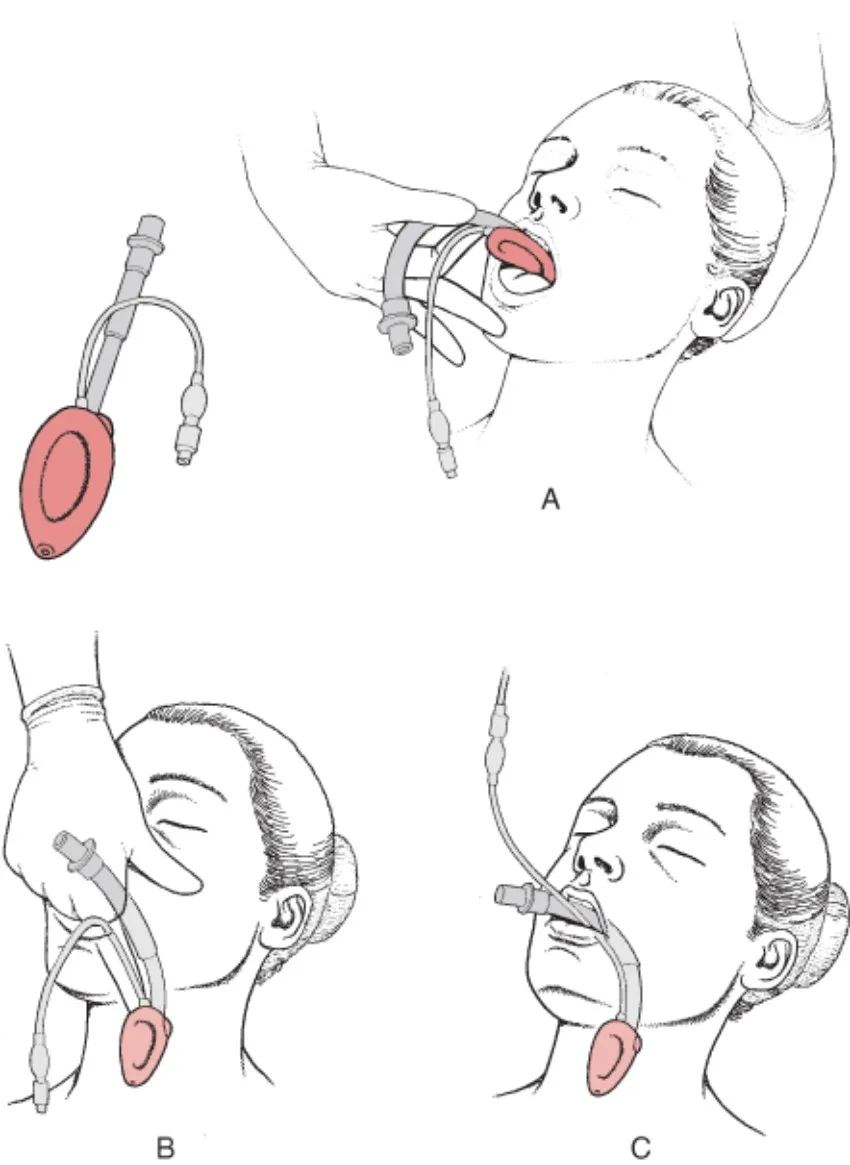
Common Usage Scenarios:
Important Notes:
• Use only by trained medical professionals.
• Check device and packaging integrity before use.
• Do not reuse disposable models.
• Avoid over-inflation of the cuff to prevent mucosal injury.